Part 2 - return to Part 1
Pathways to depression and anxiety – malnutrition, toxicity and microbiome dysregulation. (Undergrad paper, the condensed nature of the paper a function of the assigned word limit).
Humans have long referenced the gut and the brain in common lexicon, yet science is only now catching up. Gut-brain communication, or the gut-brain axis is a complex biological system that supports behaviour and physiological processes; including bi-directional neuroendocrine signalling, immune activation, gut brain cross-talk, hormonal signalling and modulation of sensory-motor reflexes. The gut microbiota has
‘emerged as a critical component potentially affecting all of these neuro-immuno-endocrine pathways’ (Foster, Rinaman, & Cryan, 2017, p. 125).
The scientific literature is quite clear that the gastrointestinal health, connecting the brain-gut-microbiome, plays a significant role in psychiatric and neurological disorders including anxiety and depression-like behaviours (Martin, Osadchiy, Kalani, & Mayer, 2018).
This paper proposes that the average Western diet is insufficient to protect the individual against the assaults of daily life. Dietary inadequacy can be attributed to fatigue, a weakened immune system, dysregulated circadian rhythms, and inducing brain fog, all of which are symptoms of anxiety and depression.
Dietary toxicity can result in hormone disruption and neurological disfunction. Dietary patterns can precede the onset of psychiatric symptoms (Rucklidge & Kaplan, 2016). Increasingly, the evidence suggests that diet can prevent depression (Sanchez-Villegas & Martinez-Gonzale, 2013; Stahl, Albert, Dew, & al, 2014; Ven de Weyer, 2006).
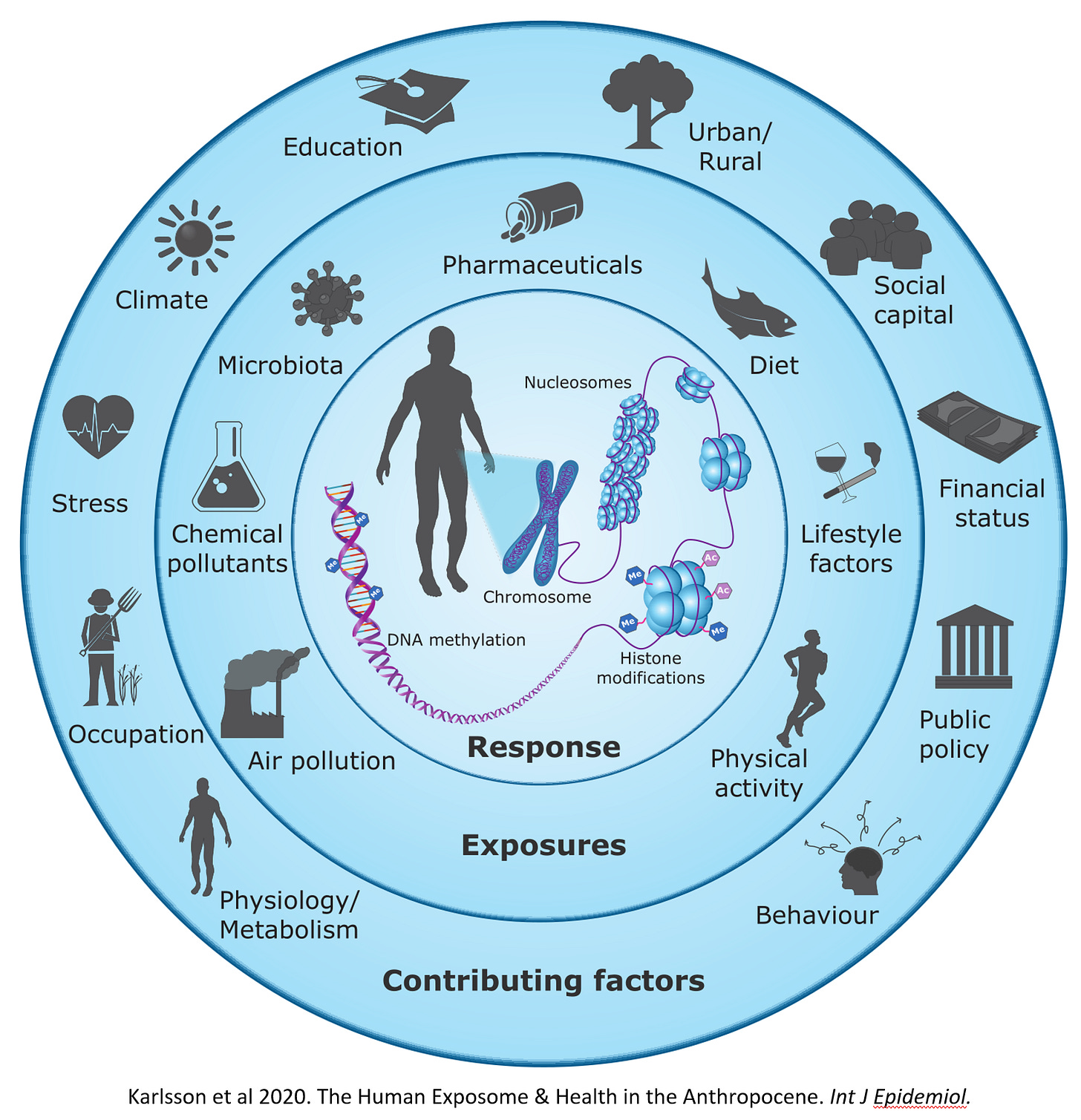
After the decoding of the genome and the shock finding that human genes amounted to a quarter of the original estimate that genes would be identified to be the predominant driver of non-communicable disease, - the post-genomic epigenetic era commenced (Brower, 2001).
Genetic expression - a function of micronutrient inputs
Scientific research has brought to light that we are not so much driven by genes but by genetic expression. This relatively new field, referred to as epigenetics, recognises that gene expression is shaped by the environment, including exposures from stress, nutrients, synthetic chemicals and dietary fibre. Epigenetic interactions do not tend to alter the DNA sequence and can be heritable across generations (Nilsson, Sadler-Riggleman, & Skinner, 2018).
The gut does not merely digest food, convert it to energy and regulate the immune system, - the metabolite chemicals produced in the microbiota are thought to be a primary mechanism in the regulation of the epigenome, influencing and driving DNA and gene activation. Where it was thought the genome contained the instructions for life, post-genomic science reveals the genome as a blueprint, with the human body a network of epigenetic ‘crosstalk’ between the bacteria contained in the gut microbiota and the human host, or epigenome enabling gene expression and function (Qin & Wade, 2018).
Diet is the most important risk factor for chronic disease (WHO, 1990).
There is broad-ranging evidence that wide swathes of the population are undernourished (Grotto & Zied, 2010) (Tobias, et al., 2006) (Tulchinsky, 2010), and yet more evidence to support that nutrition (Stanga, et al., 2007), and reducing dietary exposures from toxic chemicals (Defois, et al., 2018) improves psychological function.
Micronutrient deficiencies, particularly iron related, among adolescents are common and girls are particularly vulnerable (Christian & Smith, 2018). In addition to iron deficiency, common deficiencies are firmly associated with mental health, including magnesium (Liu & Zhu, 2018) vitamin D (Anglin, Samaan, Walter, & McDonald, 2013), copper, iodine and selenium.
Micronutrients and mental health - time for policy to reflect the data [Ed. 2022]
Vitamins and minerals play an important role in brain health, acting as cofactors in the synthesis and metabolism of neurotransmitters, which regulate our neuronal systems. (Rucklidge, JJ, Andridge R et al. 2012.)
The critical role of micronutrients as cofactors in neurotransmitter function (see also Kaplan et al., 2007) Mylniec, et al., 2015) has been widely ignored by governments, mental health support organisations and media.
A brief summary is provided here by Julia Rucklidge and colleagues. This information, from a ten year old paper, demonstrates that nutrient-brain science has been well under way for some time:
For example, thiamine (B 1 ) protects the adrenal glands from exhaustion; niacinamide (B 3 ) shunts tryptophan to serotonin; pyridoxal 5 phosphate (B 6 ) acts as a cofactor for the synthesis of gamma-aminobutyric acid, serotonin, and dopamine; methylcobalamin (B 12 ) normalizes cortisol production; ascorbic acid (vitamin C), given in higher than recommended dietary allowance values, supports adrenal function and decreases high cortisol levels; and 5-methyltetrahydrofolate regenerates tetrahydrobiopterin, which is essential for neurotransmitter formation (see Head and Kelly, 2009). Folate, vitamin B 12 , and B 6 are involved in the metabolism of homocysteine, which is a by-product of methionine metabolism. Recent research shows that there is a clear relationship between homocysteine levels and stress (Kang et al., 2005), possibly caused by a reduction in B 6 levels by acute stressors and that supplementation with B 6 may result in reductions in stress. With these physiological mechanisms, supplementation with B vitamins may be sufficient to decrease stress levels, at least in the short-term
In 2021 Julia Rucklidge and Bonnie Kaplan released The Better Brain, and I’ve written previously that I believe Rucklidge is New Zealand’s most popular professor you may never have heard of.
The Better Brain is a paradigm shifting book that should be integrated into the education and health systems - by governing bodies who claim they promote evidence based policy.
The Better Brain beautifully complements and reflects work underway in New Zealand by others to reverse metabolic diseases including type 2 diabetes. Patients with T2D often have struggle with mental illness.

Malnutrition & vulnerability to mental illness
(Back to the original paper..)
Symptoms of malnutrition correlate with depression and/or anxiety, and can include:
reduced heart rate variability (Barreto, Vanderlei, Vanderlei, & Leite, 2016; Tolentino & Schmidt, 2018; Young & Benton, 2018);
dysregulation of the hypothalamic–pituitary–adrenal (HPA) axis (Cooke, James, Landon, & Wynn, 1964; Xiong & Zhang, 2013; Young, Abelson, & Cameron, 2004);
fatigue;
sleep disruption (Peirano, Fagioli, Singh, & Salzarulo, 1989); and
irritability and loss of concentration (Stanga, Field, Stucki, Lobo, & Allison, 2007).
A dysregulated HPA axis impacts cortisol levels which drive circadian rhythms. Nutrient deficiency can elevate glucocorticoid levels (Cottrell, Holmes, Livingstone, Kenyon, & Seckl, 2012) and high glucocorticoid signalling is associated with impairment in the depressed brain (Anacker, Zunszain, Carvalho, & Pariante, 2012). HPA dysregulation may evolve in the infant from nutrient restriction in the mother (Chadio, et al., 2007) as in adverse behavioural result from a mother who may have depression, without considering the mother may also be experiencing HPA dysregulation (Essex, et al., 2011), and that this may be the result of an inadequate diet (Lesage, et al., 2009), suggesting a familial, diet related aetiology.
Finally, formula fed to infants results in less robust microbiomes (O'Sullivan, He, Haggarty, Lönnerdal, & Slupsky, 2013); antibiotics, a caesarean or natural birth and viruses can also adversely impact the microbiome gut flora (Cortese, Lu, Yu, Ruden, & Claud, 2016; Qin & Wade, 2018).
The connection of malnourishment, or undernourishment – as a broader dietary impact - to depression and anxiety is significant.
The gut-brain axis is a
‘bi-directional communicative and regulatory system involving (but not limited to) the brain and central nervous system (CNs) and the enteric environment of the gut’
(Schnorr & Bachner, 2016, p. 398).
The gut microbiota includes microbial cells, including human, bacterial, fungal, metabolites and neuroactive chemicals. The enteric nervous system interconnects the brain and gut via neural pathways including the vagus nerve and the bloodstream (Schnorr & Bachner, 2016).
Gut microbiota is mediated by the interactions of nutrients, fibre, and synthetic chemicals which impact production of the neural, endocrine and immune systems influencing brain and behaviour and risk of health or disease (Cryan & Dinan, 2012; Levy, Kolodziejczy, Thaiss, & Elinav, 2017).
Microbiota in the gut synthesise vitamins and mediates the inflammatory response to manage stress (Qin & Wade, 2018); synthesises and metabolises hormones (including serotonin) and amino acids (tryptophan), maintaining the immune system.
Scientists have built a compelling body of evidence, demonstrating that gut health is key to mental health (Dickeson, Severance, & Yolken, 2016; Foster, Rinaman, & Cryan, 2017; Rieder, Wisniewski, Alderman, & Campbell, 2017).
Western-style diets are low in microbiota-accessible carbohydrates, and prebiotic dietary fibre variability is important for promotion of microbial diversity (Makki, Deehan, Walter, & Bäckhed, 2018).
Environmental exposures & the gut
The microbiome is adversely impacted by exposures from the surrounding environment. Chemical pollution, whether in the foetus, child or the natural environment, is a waste product sequestered. The polluted child is Moore’s ‘cheap nature’ (Moore, 2015).
Environmental chemicals also play a role in the pollution and disruption of the gut microbiome, and the challenges to gut health that result from chronic, low level lifetime exposure. Chemicals can drive gut inflammation, ribosome, translation and nucleic acid binding (Defois, et al., 2018) and mimic and damage hormone functioning (Trasande, 2019).
While organic diets have been propagandized as an elite privilege, before the twentieth century most diets were organic.
Chemical regulators have not kept pace with the evidence that chemicals at very low doses can be harmful at endocrinological levels (Slama, et al., 2016). Regulator have avoided considering the toxicity of mixture effects (Kortenkamp A., 2014; Kortenkamp & Faust, 2018).
Scientists have documented the neurotoxic effects following chronic exposures to environmental chemicals at low levels (Barreto, Vanderlei, Vanderlei, & Leite, 2016). The health based and neurological cost to society from endocrine disrupting chemicals has been estimated to be 2-3% of GDP (Attina, et al., 2016). Children are particularly vulnerable (Landrigan & Goldman, 2011).
The significant presence of comorbid health conditions (Brown, Harris, & Eales M.J.; RANZCP, 2016, p. 21; Everett, Mahler, Biblin, Ganguli, & Mauer, 2014) including metabolic syndrome (Dunbar, et al., 2008) reinforce the fact that physiological health is intricately connected to mental health.
Over 60% of depression and anxiety patients present with gastrointestinal dysfunction, for example, irritable bowel. When the intestinal barrier is compromised, inflammatory molecules entering the bloodstream and immune system can set off a cascade of depressive effects. (Liu & Zhu, 2018)
Sociological theory and the black box of the human body
The brain as ‘problem’ has been a convenient strategy, as without a ‘cure’ or ‘solution’, psychiatric medicalisation which prioritises mechanistic pathways to symptom suppression, inevitably gifted the pharmaceutical industry a Pandora-like never-ending box of never-ending so-called solutions to problems of disease and disability.
Psychiatry as a solution has long been criticised by sociologists (Pilgrim & Rogers, 2005). Common sociological criticisms include the fact that drugs marketed for mental illness are not better than a placebo, and in fact can be worse, albeit with a slew of detrimental adverse effects, including risk of iatrogenic harm.
The boundless and gendered nature of the symptom categories, particularly in relation to the medicalisation of children has been roundly criticised (Cohen, 2016; Whitaker & Cosgrove, 2015). Authors have noted the capture of health professionals and other actors to the biomedical/psychiatric industrial complex (Gomory, Wong, Cohen, & Lacasse, 2011).
Patent management, including strategies to extend patent protection via new product applications remains part of the business model (Whitaker & Cosgrove, 2015) as without patents,
‘the coin of the proprietary realm’
the pharmaceutical industry cannot profit (Owen-Smith, 2007, p. 70).
Problems pervade medicalised narratives. Only fifty per cent of depressed patients might be responsive to available treatment (Menard, Hodes, & Russo, 2016) or as little as three per cent would ‘stay well,’ or only the very ill would express a benefit (Whitaker & Cosgrove, 2015). Drug development is often hampered by treatment-resistance and problematic side effects (Hillhouse & Porter, 2016; Papakostas, 2008).
The sociological focus on stress and trauma.
Sociological researchers of stress theory suggested adversity, including unhappy marriages and social exclusion to create chronic stress that impacts mental health (Aneshensel, 1992). Adverse experiences in childhood and adulthood, inadequate social infrastructure and comorbidities increase risk for depression and anxiety but cannot be identified as causal (Brown & Harris, 1993).
There is not strong evidence that negative events and chronic strains necessarily lead to mental impairment, as many people experiencing severe stress maintain their health (Thoits, 1999, p. 126). There are very few sociological accounts exploring the microbiome, diet and mental health.
‘Social theory has often rendered the human body tabula rasa, ‘bracketing’ it as a black box’ (Palsson, 2017).
There appears a sociological reluctance to grant status to biological and genetic factors that underlie disease and illness. Researchers propose this has been based on a cultural reluctance to devalue social factors (Timmermans & Haas, 2008). Authors have noted the inclination of the sociological sciences to exclude biology and disease in research of health and illness, instead bracketing the biological ‘bedrock of disease’ (Timmermans & Haas, 2008, p. 660).
Hannah Landecker and Aaron Panofsky identified distinct opportunities in the sociological sciences to engage with the new scientific field of epigenetics. Environmental epigenetics researches genetic regulation and response to nutrition, pollution and stress and brings to public attention the transgenerational of adverse exposures, either to trauma, diet or chemicals.
A mother that is malnourished or toxic may not only deplete and alter genetic expression in the foetus, but in the egg of a daughter foetus. Three generations can be harmed at once. This should profoundly change policy and regulatory approaches to risk, promoting discussion and policy that draws into consideration notions of epigenetic social responsibility (Landecker & Panofsky, 2013).
Western food cultures are dominated by ultraprocessed food consumption
While severe and sustained trauma can predict mental illness, I submit that diet is a significant factor in the majority of cases presented to doctors in Western practice today. It is well known that are familial patterns of disease (Levinson, 2005).
Western culture has devalued whole, chemical free food and the fallout from this new malnutrition and toxicity is broadly manifesting itself, and this can be most closely observed in household, or familial environments (Levinson, 2005).
Ivan Illich drew attention to a new kind of malnutrition consisting of undernourishment and toxicity, and noted that food air water and the level of equality are dominant factors in health outcomes (Illich, 2000, pp. 18-20).
This contention may can help explain why groups are more likely to be diagnosed with mental illness; and add new insight into understandings regarding the ways by which socio-economic status and class plays a part in depression and anxiety (Goodman, Slap, & Huang, 2003; Holllingshead & Redlich, 1958; Ostler, K., et al., 2001; Srole & Langer, 1962); but also explain why wealthy populations can be vulnerable (Luthar & Latendresse, 2005).
Many relatively wealthy and middle-class households have poor diets and food addiction can play a central role in food choice no matter the socio-economic status of the family (Lustig, 2017).
Structural strain hypothesis
The structural strain hypothesis sheds light on the methods by which hard-economic times, stressful events and traumatic events contribute to mental illness; and recognises that disadvantaged groups are more likely to experience greater strain and are more likely to be diagnosed with a mental illness (Thoits, 1999). Structural strain theory may explain the cultural shift (as social patterning) to cheaper, low protein processed food diets, and the greater probability that poorer families will consume high calorie, empty nutrient diets.
As such, dietary stress theory as a major driver of depression and anxiety, represents a sociological opportunity to drill down from the macro to witness the impact of micro and familial manifestations of neoliberal culture, and incorporate the black box of malnutrition and toxicity as (dietary) stress theory.
Mental health resilience is buffered by microbiome resilience, and dietary habits are constants in family environments.
The issues around dietary stress theory reflects Durkheimian social problems and Marxist concepts and concerns.
How we eat, one of the most fundamental practices that has absorbed man since cell division, has fundamentally changed (Cordain, et al., 2005) This contestation is sociological, as it directly challenges capital and culture.
The cultural and normative approach to cooking has been replaced by access to preassembled ultraprocessed food. Marxist themes resonate, as the cartel-like giants owning and shaping the food supply directly reflect Marx’s concerns with ownership of the material relations of production and his accusation that economic power not only impact the legal and political but may manipulate the ideological shaping of society to impact, as Marx explained ‘the social consciousness’ (Cohen, 2016, p. 29).
Sociologist Bruce Cohen maintains the psychiatric professions act as profoundly political ‘agents of social control’, serving a capitalist agenda. In this environment encroaching medicalisation has served a political and economic purpose, not only garnering profits for the pharmaceutical industry, but controlling deviant populations and assuring reproduction of norms and values (Cohen, 2016).
Perhaps the psychiatric profession has an additional, largely unrecognised role, to suppress the fallout from malnutrition and toxicity and ensure the neoliberal agro-industrial machine continues unabated.